In an era where technology transcends borders and healthcare innovation defines global competitiveness, Bangladesh is quietly but steadily scripting its own success story. The medical equipment and device sector—once limited to import dependency—is now emerging as a dynamic player on the world stage. Under the strategic coordination of the Medical Equipment & Device Manufacturer and Exporter of Bangladesh (MEDMEB), the country’s industry is entering a transformative phase driven by Foreign Direct Investment (FDI), Original Equipment Manufacturing (OEM) partnerships, and cross-border technology sharing.
From Local Manufacturing to Global Mindset
The turning point came when Bangladeshi manufacturers realized that sustainability in healthcare production required more than just local supply—it needed global thinking. The vision was not only to assemble products but to become an integral part of the international medical device value chain.
Companies like PROMIXCO Healthcare, JMI Group, and Bangladesh Multifibers Co. Ltd. have become symbols of this change. With state-of-the-art production facilities, ISO certifications, and DGDA registration, they are aligning with EU MDR, US FDA, and ASEAN standards. These efforts are turning Bangladesh from a market of consumption into a hub of collaboration.
The shift is deliberate. No longer content to merely distribute imported goods, Bangladeshi manufacturers are developing R&D partnerships, OEM agreements, and technology licensing deals with companies from Japan, South Korea, Germany, and the UAE. The aim is simple but bold: to transform “Made in Bangladesh” into a global mark of reliability and innovation.
The Rise of FDI in HealthTech Manufacturing
Foreign investors are taking notice. According to MEDMEB’s 2025 report, FDI in HealthTech grew by 22% in just one year. The surge comes as the Bangladesh Investment Development Authority (BIDA) and the Directorate General of Drug Administration (DGDA) streamline processes for overseas investors. These include simplified registration for joint ventures, flexible tax incentives, and preferential access to export-processing zones for healthcare manufacturing.
Japan and Germany have led the wave with pilot projects in automation, material engineering, and sustainable sterilization systems. The UAE and South Korea have followed with commitments in hospital infrastructure and smart medical devices.
Bangladesh’s competitive advantage lies in three pillars:
1. Cost Efficiency: Production and labor costs remain 40–50% lower than regional competitors.
2. Regulatory Reform: MEDMEB’s collaboration with DGDA has ensured alignment with WHO’s medical device regulatory framework.
3. Market Access: Duty-free export channels under GSP and South-South trade networks allow seamless movement to 30+ countries.
The FDI flow is not merely financial—it’s transformational, bringing in expertise, automation technology, and managerial excellence.
OEM Partnerships: Building Bridges, Not Barriers
One of the most powerful enablers of industrial modernization is OEM collaboration. Through OEM deals, foreign companies license their technology or designs to local manufacturers, who then produce and distribute devices under strict quality supervision.
In 2025, Bangladesh signed 14 new OEM and co-branding agreements, spanning sectors from hospital furniture and diagnostic kits to patient monitoring and physiotherapy equipment.
Notable ventures include:
– A Japan-Bangladesh consortium producing smart ICU beds equipped with IoT sensors for patient movement and vitals tracking.
– A Germany-Bangladesh partnership focusing on modular sterilization units with CE compliance.
– UAE-based investors working with local partners to manufacture infusion pumps and nebulizers for the Middle East market.
These partnerships go beyond profit. They represent technology democracy—a model where innovation flows from one part of the world to another without losing local relevance. MEDMEB’s OEM Facilitation Desk now serves as a one-stop platform for matching foreign innovators with Bangladeshi producers who can deliver at scale.
Technology Sharing and the “TechBridge” Program
The MEDMEB “TechBridge Program,” launched in mid-2025, has become the linchpin of cross-border innovation. The initiative connects Bangladeshi engineers and R&D units with global universities, think tanks, and manufacturers. Through knowledge transfer workshops, digital platforms, and co-lab prototypes, the program encourages shared R&D, material science collaboration, and clinical validation exchange.
Key collaborations already underway include:
– MIT-MEDMEB Digital Diagnostics Exchange, focusing on AI-assisted pathology tools.
– Tokyo Tech Partnership, promoting precision engineering and sensor calibration.
– Stanford–Dhaka Biomedical Consortium, exploring low-cost wearable devices for chronic disease monitoring.
The message is clear: Technology sharing is not an option; it is the foundation of global competitiveness. MEDMEB is ensuring that Bangladeshi innovators don’t work in isolation but as part of an international ecosystem of healthcare transformation.
Policy and Regulation: Building the Framework for Tomorrow
Every industrial revolution needs a policy backbone. MEDMEB, in collaboration with DGDA and the Ministry of Industries, is drafting the National OEM Partnership Framework (2026–2030) and the FDI Facilitation Roadmap for HealthTech Manufacturing. These policy documents aim to:
– Introduce a unified certification and inspection regime.
– Simplify import of raw materials and spare parts for export-oriented manufacturers.
– Establish industrial zones dedicated to medical device R&D and assembly.
– Integrate medical devices into the National Industrial Policy Amendment 2026.
Once approved, these frameworks will anchor Bangladesh’s long-term ambition to be a regional manufacturing hub. MEDMEB’s advocacy ensures that government incentives match global expectations, paving the way for sustained foreign engagement.
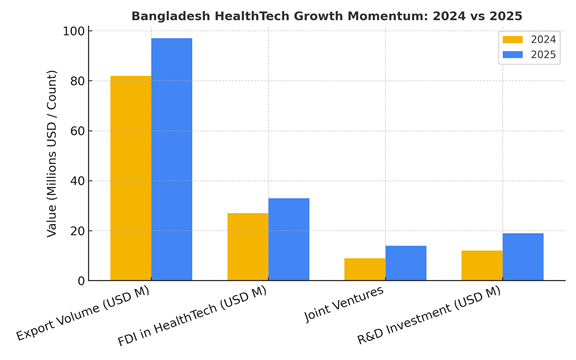
Export Momentum: Numbers that Speak
The export graph tells a compelling story.
Indicator | 2024 | 2025 (YTD) | Growth
———–|——|————-|——–
Export Volume (USD) | 82M | 97M | +18%
FDI in HealthTech | $27M | $33M | +22%
Joint Ventures Registered | 9 | 14 | +55%
R&D Investment (Private) | $12M | $19M | +58%
Bangladesh’s medical device exports now reach over 30 countries, with major shipments going to India, Nepal, the UAE, Egypt, Kenya, and Poland. The hospital furniture and diagnostic device categories lead the way, accounting for 62% of total export value.
Conclusion: Collaboration is the New Currency
In the end, Bangladesh’s rise in the global medical device sector isn’t about who invests or who manufactures—it’s about who collaborates best. The new global economy rewards openness, partnership, and shared innovation.
FDI brings capital. OEM brings technology. MEDMEB brings unity. Together, they redefine Bangladesh’s industrial destiny.
As Chairperson Mousumi Islam aptly said, “With 18% export growth and a 22% FDI rise, Bangladesh’s medical device industry is entering its global decade.”
From Dhaka to Dubai, from Tokyo to Maryland—the world is watching a new chapter unfold: Bangladesh is not waiting for the future of healthcare manufacturing. It’s building it.
